The liver is one of the largest organs in the body, with a weight of approximately 3 pounds. Some of its vital functions include the filtration of blood to remove toxic substances from the body, the production of bile which helps with digestion, and the regulation of fat metabolism. Fats or lipids from the diet are taken up by the liver and processed into fat-carrying proteins called lipoproteins. These lipoproteins are released into circulation to fuel tissues that require energy. However, when there is a positive energy balance, for example due to overeating and/or a sedentary lifestyle, liver cells increasingly store lipids. This can result in metabolic associated fatty liver disease (MAFLD, formerly known as NAFLD), characterized by a liver fat content above 5%. When fat keeps accumulating in the liver, chronic inflammation ensues and the liver progresses to a stage called metabolic associated steatohepatitis (MASH, formerly known as NASH). There are currently no FDA-approved drugs available to treat MASH. At this stage, patients are at risk for developing a type of liver cancer called hepatocellular carcinoma (HCC). HCC is the primary cause of liver cancer in the US, affecting more than 30,000 individuals per year. Progression from a healthy liver to MAFLD, MASH, and HCC is shown in the Figure below.
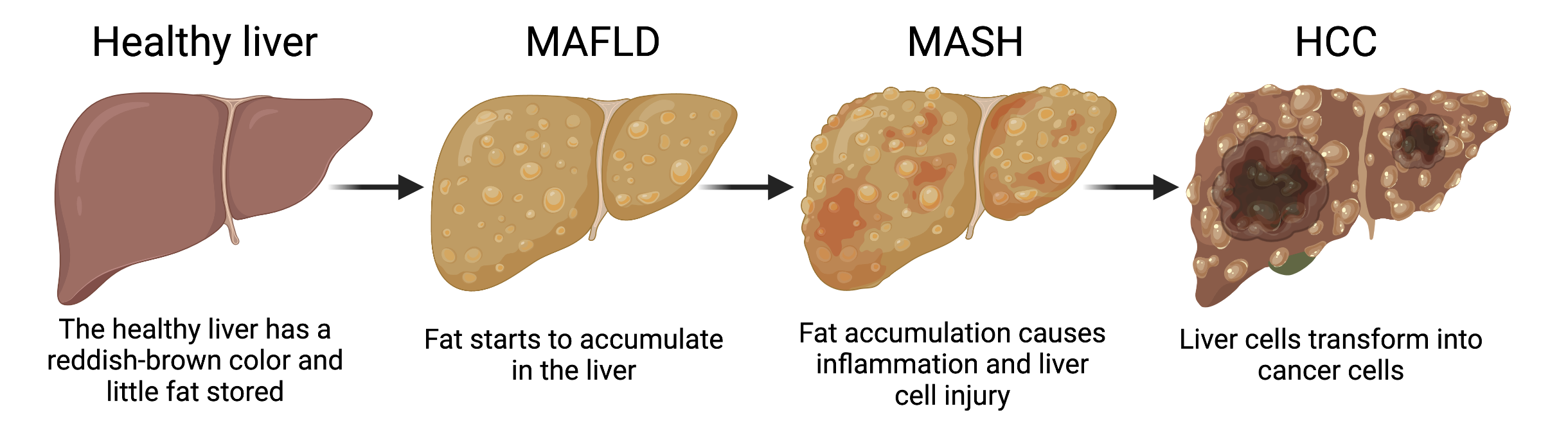
The different stages of fatty liver disease progression – from a healthy liver, to metabolic associated fatty liver disease (MAFLD), metabolic associated steatohepatitis (MASH), and eventually hepatocellular carcinoma (HCC). Figure created with Biorender.com.
Although MASH is the leading cause of HCC, the mechanisms of how MASH predisposes to HCC tumor formation are largely unknown. The research from Columbia postdoc Xiaobo Wang and colleagues tries to fill this knowledge gap. Dr. Wang investigated TAZ; a gene regulator that was found to be increased in MASH livers. He fed experimental mice with a diet containing high sugar, fat, and cholesterol (the equivalent of human “fast food”), to induce MASH development. Then, he diminished TAZ expression in the liver by using a viral-mediated gene delivery system, by which an engineered virus enters mouse liver cells to specifically turn off the TAZ gene. Silencing of the TAZ gene largely prevented the development of tumors in MASH liver, indicating that TAZ is an important player in MASH-HCC progression.
Dr. Wang continued his research by investigating how TAZ could enable the liver cells to turn into tumor cells. He focused on DNA damage, a process which is important in HCC development, and found clear indications of damaged DNA in the livers of mice and humans with MASH. Most importantly, silencing of TAZ prevented an increase in the DNA damage, suggesting that TAZ promotes genomic instability in liver cells. Since the buildup of oxidative stress within cells is an important cause of DNA damage, Dr. Wang next looked at a specific indicator of oxidative DNA damage. Indeed, this indicator was increased in MASH and decreased with TAZ silencing. He then measured various oxidant-related proteins to find out how TAZ could promote oxidative DNA damage. He discovered that Cybb, a gene involved in the formation of harmful reactive oxygen species, is involved in TAZ-induced liver cancer. Together, these findings show a TAZ-Cybb-oxidative DNA damage pathway (see Figure below) that creates malignant liver cells and promotes the progression from MASH to HCC. This work has been published in the prestigious Journal of Hepatology.
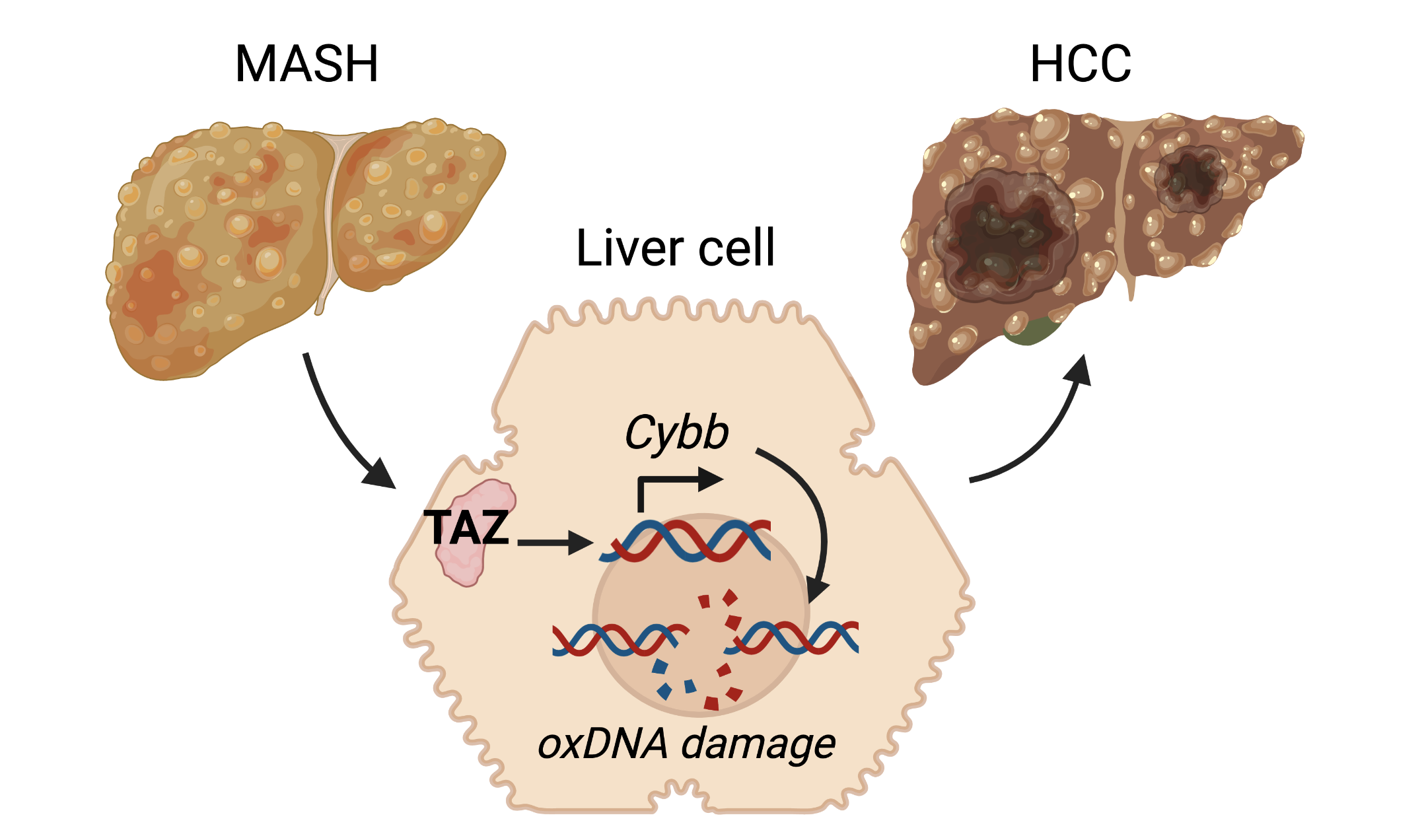
Metabolic associated steatohepatitis (MASH) liver cells highly express a protein called TAZ, which promotes the Cybb gene which is involved in production of reactive oxygen species. These reactive oxygen species induce oxidative DNA damage, which transforms healthy liver cells into tumor cells and thereby promotes progression to hepatocellular carcinoma (HCC). Figure adapted from Wang et al. J Hepatol 2021, and created with Biorender.com.
The new pathway that Dr. Wang and colleagues discovered suggests that TAZ-based therapy could prevent MASH-HCC progression in humans with fatty liver disease. Such a therapy could have a big clinical impact, since it is estimated that about 12% of US adults have MASH. However, a limitation to MASH therapies is that the disease is often asymptomatic and difficult to identify until the late stages of the disease. Aside from focusing on treatments that reduce MASH progression after a large part of the damage has already occurred, our society should increasingly focus on preventative strategies. MASH is often caused by lifestyle-related factors, and risk of MASH can be significantly reduced by maintaining a healthy weight, eating a healthy diet and exercising regularly. Especially in the US where MASH prevalence is expected to increase by 63% by 2030, raising awareness of the importance of living healthy to prevent liver disease and liver cancer is paramount.
Reviewed by:
Sam Rossano, Vikas Malik, Molly Scott