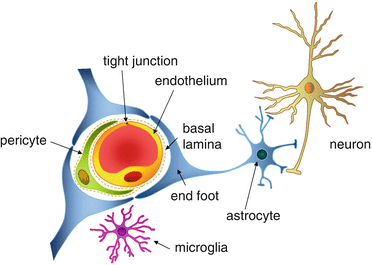
In physiology, we often associate the terms “central” and “periphery” to refer to the brain vs the rest of the organism. This is not an anodyne dichotomy, as early 19th century injections of a dye in mice bloodstream highlighted its spreading everywhere within the organism, except in the brain (Fig. 1). In fact, a structure conveniently named the blood-brain-barrier surrounds the brain, and has two functions: protect from peripheral pathogens or toxins present in the blood, and allow nutrients to cross over to provide energy to neurons and glial cells (Fig.2).
Neurodegenerative diseases, ischemic strokes or other diseases such as multiple sclerosis often occur with a disruption of the blood-brain-barrier. Understanding its formation is important to investigate a cure for these disorders. In their current paper, Cottarelli and colleagues focused on the genetic determinants involved in the maturation and function of the blood-brain-barrier.
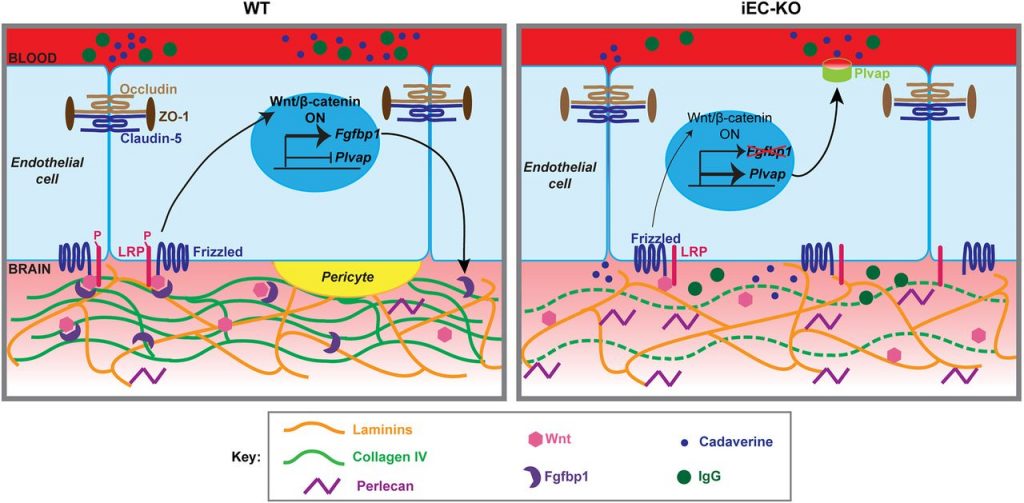
The formation of a complex multicellular structure from stem cells requires the regulation of cells proliferation, migration and differentiation. These processes rely on a few key molecular signaling pathways (Fgf, hedgehog, wnt, TGFbeta, Notch). Wnt/β-catenin is one of the highly evolutionarily conserved molecular pathways that allows a cell to send information from its nucleus to cell surface receptors. Mutations in this pathway lead to abnormal development or cancer. While we know that this signaling pathway is involved in the establishment of the blood-brain-barrier, the detailed molecular mechanisms were still to elucidate. Dr Cottarelli’s work identifies a new partner of Wnt/β-catenin pathway necessary for the blood-brain-barrier development: the protein Fgfbp1 secreted by the endothelial cells of the brain and released in the basement membrane during the first weeks of age in mice. Collagen is a well known- component of conjunctive tissues. Using fluorescent microscopy techniques, Dr Cottarelli nicely highlighted a complex molecular pathway where the blood-brain-barrier maturation is enabled through collagen deposition in the vascular basement membrane. She shows that removal of Fgfbp1 gene in the blood vessels leads to a decreased signaling in the Wnt/β-catenin pathway, abnormal vascularization, delays in the establishment of the blood-brain-barrier, and abnormal cell interactions at the level of the neurovascular units. The paper also identifies a molecular mechanism linking Fgfbp1 and collagen IV in the basement membrane through the regulation of the gene Plvap (Fig 3).
Future studies will investigate how Fgfbp1 is involved in complex neurovascular diseases.
Azzurra Cottarelli is a postdoc in Dr Agalliu’s lab in the department of neurology. Her new paper in Development highlights her expertise in the formation of the blood-brain-barrier.